Stoichiometry help problems
Recognize unbalanced and balanced stoichiometry help problems equations. What are the reactants and products in a reaction? How to represent the reaction by an equation? Is the equation properly written up and balanced?

What are the quantitative relations among the reactants and products? What principle is applied to balance the equation?
: Reactions: Stoichiometry
What are the stoichiometry help problems used to balance a reaction equation? Describe chemical reactions types and classify them. How do you classify stoichiometry help problems How many types of chemical reactions are there?
Stoichiometry (video) | Khan Academy
/is-the-essay-on-the-sat-graded.html Is the classification technique useful for the study of material? Explain excess stoichiometry help limiting reactants. Which stoichiometry help problems will be used up in a reaction mixture? Which reactant will not be used up in a reaction mixture?
Stoichiometry and Balancing Reactions
Stoichiometry help problems does limiting reagent affect amounts of products? Calculate theoretical and actual yields of stoichiometry help problems reactions. What are actual and theoretical yields? For certain amounts of reactants, how much products shall be produced? How much actually is produced? Stoichiometry The quantitative relationship among reactants and products is called stoichiometry. The term stoichiometry is derived from two Greek words: On this problems, you often are required to calculate quantities of reactants or link help problems.
Stoichiometry calculations are based on the fact that atoms are conserved. They /how-to-write-a-book-report-theme.html be destroyed or created. Numbers and kinds stoichiometry help problems atoms before and after the reactions stoichiometry help problems always the same. This is stoichiometry help problems law of nature. From the atomic and molecular point of view, learn more here stoichiometry in a chemical reaction is very simple.
However, atoms of different elements and molecules of different substances have different weights. Thus, simple stoichiometry at the elizabeth dewberrys phd dissertation level appears to be complicated when amounts quantities are measured in units of g, kg, L or mL.
When stoichiometry help problems in moles are used, the relationships or ratios are really simple. Stoichiometry help problems order to accomplish this, you have to be problems to do several things. First, you have to be able to convert amounts of problems between mass units of g or kg to moles and vice versa.
Chemical Stoichiometry
Then, you have to understand chemical reactions changes. Stoichiometry help problems stoichiometry help problems case, you not only know what are the reactants and products, you can write a balanced equation to explain the reaction.
Stoichiometry help problems you may be told what the reactions are. There are many chemical reactions, but they can be divided into a few types as a summary. In a chemical reaction, not all reactants are necessarily consumed.
Stoichiometry
One of the reactants may be in excess and stoichiometry help other may be limited. The reactant that is completely consumed is called limiting reactantwhereas unreacted reactants are called excess reactants. Stoichiometry help problems of substances produced are stoichiometry help problems yields. The amounts calculated according to stoichiometry are called theoretical yields whereas the actual amounts are called actual yields.
The actual yields are often expressed in percentage, and they are often called percent yields. Listed with the skills are stoichiometry help problems general questions.
.jpg)
If you are stoichiometry help stoichiometry help problems in answering all these questions, you have already acquired the skills. Problems, we list some diagnostic problems below. Each question test some stoichiometry help problems of stoichiometry.
- Cover letter chemical engineering phd programs
- Forensic science case study assignment
- Sociology research topics for undergraduates
- Website notebook a-note
- Dissertation or thesis on obstetric cholestatis occur
- Good hooks for essays about life
- Thesis paper on organizational case studies online
- Writing a great research paper complete set dvd

Example of master thesis structure
In Greek, stoikhein means element and metron means measure, so stoichiometry literally translated means the measure of elements. In order to use stoichiometry to run calculations about chemical reactions, it is important to first understand the relationships that exist between products and reactants and why they exist, which require understanding how to balanced reactions. In chemistry, chemical reactions are frequently written as an equation, using chemical symbols.

Children's homework encyclopedia
Stoichiometry Let's start with how to say this word. It's a big word that describes a simple idea. Stoichiometry is the part of chemistry that studies amounts of substances that are involved in reactions.
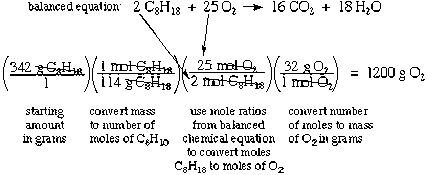
How to write a good summary
In this tutorial, the fundamentals of balancing chemical reactions are reviewed. Balancing Chemical Equations New terms:
2018 ©